Background:
Multiple Myeloma (MM) is the second most common hematologic malignancy and remains incurable. Daratumumab (dara) is a potent anti-CD38 monoclonal antibody used for MM treatment, but responses are heterogeneous, and resistance is inevitable. There is hence an urgent need for biomarkers to predict the response to dara. We hypothesized that gene expression profiles (GEP) of tumor subpopulations could be used to predict the response to dara-based therapy.
Methodology:
We used the Enhanced Single-Cell Analysis with Protein Expression (ESCAPE) RNA-Seq platform (Singleron Biotechnologies) to simultaneously profile the gene and protein expression of plasma cells from the bone marrow of 15 MM patients prior to treatment with dara-based regimens. These data were combined with publicly available single-cell data from Cohen et al. 2021, resulting in a total of 32 samples. Response was defined based on the international myeloma working group (IMWG) criteria, considering patients achieving a very good partial response (VGPR) or better as “responders,” while those achieving a partial response (PR) or worse were deemed “non-responders”.
The data were split into 23 training samples (15 responders and 8 non-responders) and 9 validation samples (3 responders and 6 non-responders), each set containing a combination of cases from the two sources of single-cell data. An interpretable machine-learning model was then built to predict the response of each individual cell. The median response score for each sample was used to predict the patient-level response.
The Shapley Additive Explanations (SHAP) values from the dara response prediction model were then used as a starting point for creating a general prognostic model. The 200 most influential genes from the dara response prediction model were used to create a linear response model based on the TCGA (MMRF-Compass) bulk RNAseq dataset. The bulk RNAseq dataset was split 80:20 into training and test datasets. Results from the test dataset are presented here.
Results:
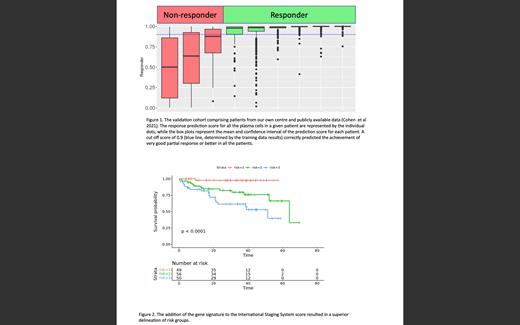
We analyzed single-cell data from a total of 13,094 plasma cells from 32 BM samples. These included 17 cases of relapsed MM described by Cohen et al., who were treated with dara, carfilzomib, lenalidomide, and dexamethasone. The patients treated at our centre comprised 7 cases of relapsed MM treated with dara, thalidomide, and dexamethasone, and 8 samples from patients with newly diagnosed MM treated with dara, bortezomib, and dexamethasone on clinical trials. All 9 patients in the test dataset had their responses correctly predicted (Fig 1). A separate cohort of non-dara-treated patient samples was also tested using the same model, and the response prediction was not accurate (AUC=0.67), suggesting that this model has specificity for predicting response to dara and is not a general response classifier (data not shown).
Based on the SHAP values from the dara response prediction model, we developed a prognostic model from bulk RNAseq data consisting of four genes. A combination of this four-gene signature plus the international staging system (ISS) score resulted in a superior prediction of overall survival compared to the ISS alone with a hazard ratio of 13.5 compared to 2.5 for the ISS alone in the same dataset (Fig 2).
Conclusions:
Combining single-cell RNA sequencing with machine-learning methods may have value in predicting response to immunotherapy in MM. We propose that gene signatures derived from single-cell data may augment clinical decision-making for determining treatment allocation. We also show that these signatures may be combined with existing prognostic scores, resulting in improved delineation of risk groups. Furthermore, the applicability of our gene signature to a bulk gene expression dataset enhances its clinical relevance. Our study demonstrates the power of single-cell omics to identify novel predictors of response to therapy and prognosis in MM, which may be translated into clinical use.
Disclosures
Scolnick:Singleron Biotechnologies: Current Employment. Xu:Singleron Biotechnologies: Current Employment. Nagarajan:BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Astrazeneca: Honoraria, Membership on an entity's Board of Directors or advisory committees; DKSH/Beigene: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: The Trial was supported by funding to IMF/AMN who were the sponsorswere the Sponsors. De Mel:Pfizer: Other: advisory board.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal